|
So, if 0.99753 g of water occupies 1 mL, then 27.8474 g of wateroccupy what volume? Things to watch out for Combining Mathematical operations/order of operations.Įxact numbers (definitions, things that can be counted, the 1 in numbers like 19.6 g/cm 3)įrom the density of water we know that 1 cm 3 weighs0.99753 g, or you could say 0.99753 g water occupies 1cm 3. multiplication/division the result of multiplying two numbers should have the same number of significant figures as the number with the fewest significant figures. addition/subtraction the result of adding two numbers together should be precise to the place that corresponds to the least precise measurementī. Determining the number of significant figures that shouldremain after performing mathematical manipulations with thenumbers. in 0.0560 the leading zeros are not significant, but the trailing zero is.Ģ. 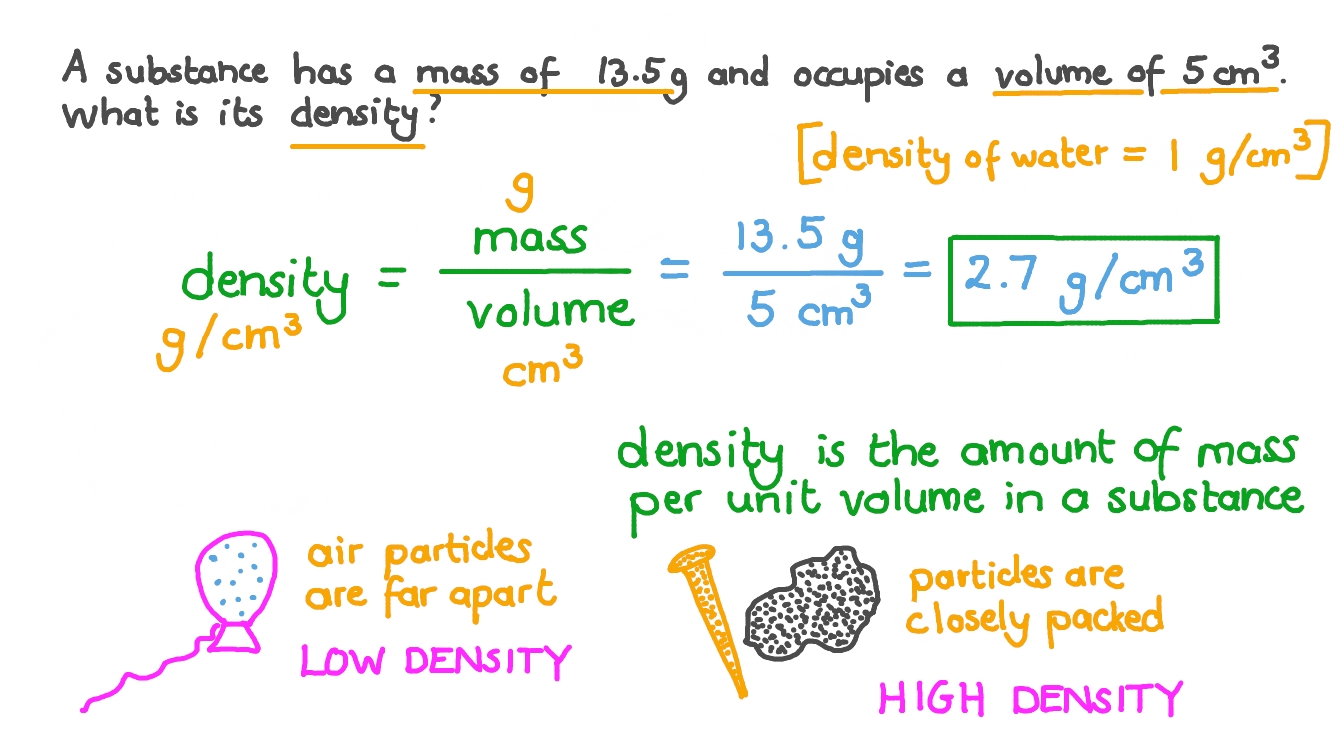
they occur after a non-zero number in a number whose absolute value is < 1. they occur before and after the decimal point of a number whose absolute value is > 1 e.g. Determining thenumber of significant figures in a measurement. Should I use 1.00 g/cm 3 as the density? No, use thedensity of water at the temperature of the experiment should be used.At 23.0 ☌ the density of water is 0.99753 g/cm 3.ġ. 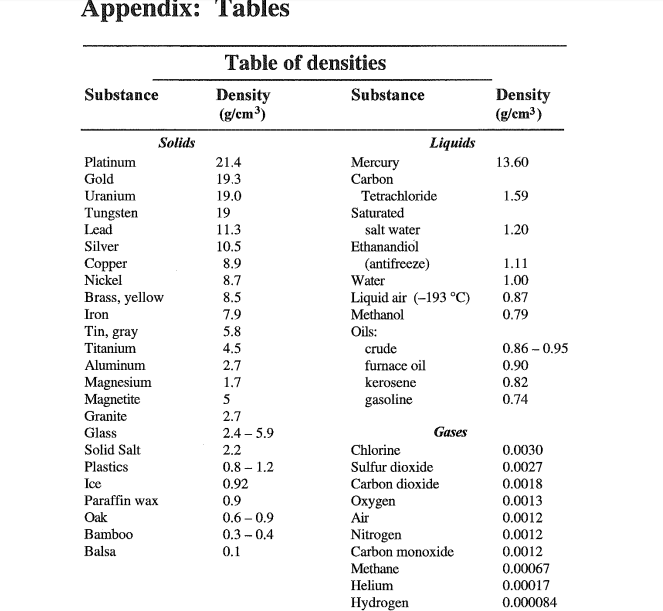
The rationale: if I know the mass of the water in the flask I caneasily convert it to the volume of the water. At 23.0 ☌ the density ofwater is 0.99753 g/cm 3įill with water, stopper, wipe, and weigh flaskĭetermine the volume of the flask using the density of water Significant Figures and the Determination of theVolume of a Picnometer (the flask)ĭetermine the volume of a flask.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
